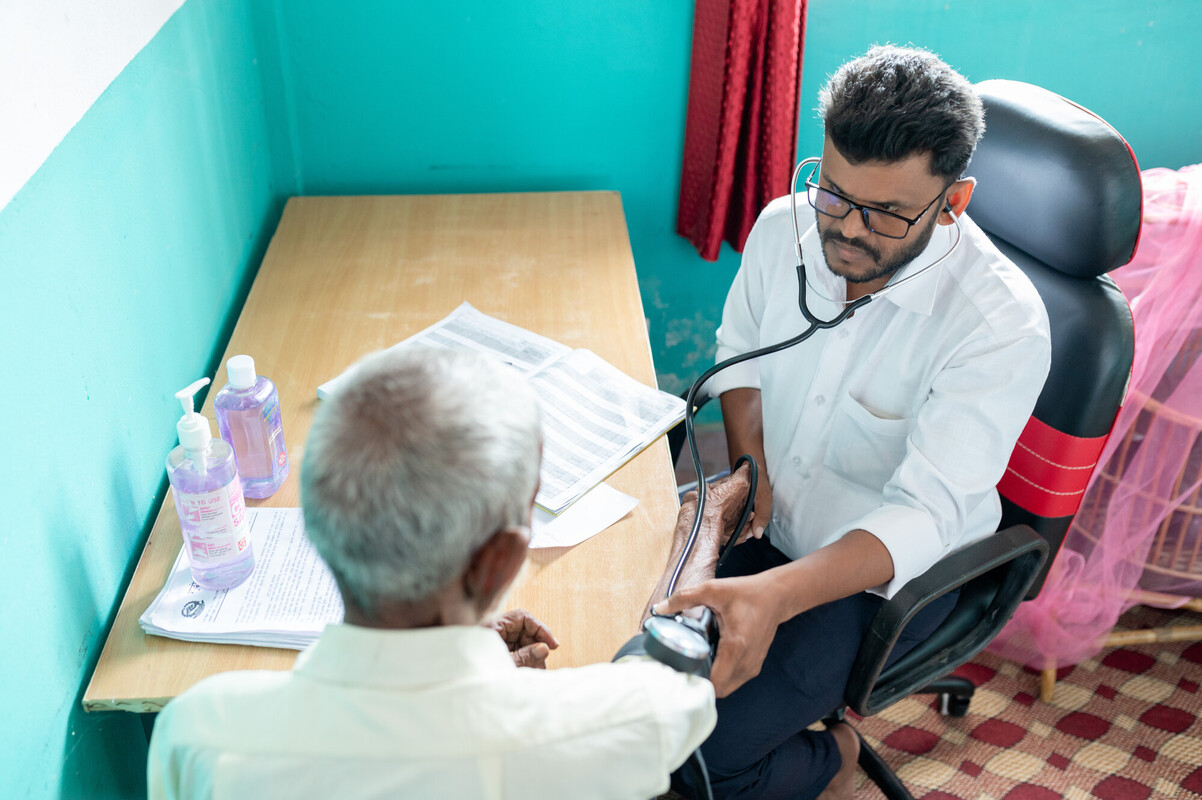
Globally, an estimated 214 million women in developing countries have an unmet need for safe and effective contraception. Hormonal contraceptive implants, introduced more than 30 years ago, are one of the most effective family planning methods developed to date. Despite their strong safety profile and popularity, their high cost has been a barrier in many countries until recently.
From 2007 to 2018, the Sino-implant (II) initiative was at the forefront of helping to increase access to affordable, high-quality implants in resource-limited settings. As a long-acting family planning method, implants are ideal for women living in countries where access to health services is limited.
Sino-implant (II), which is marketed under the global brand name LevoplantTM, is a highly effective, low-cost, subdermal contraceptive implant composed of two thin, flexible, silicone rods. Each rod contains 75 mg of levonorgestrel, a synthetic progesterone. The implants are inserted under the skin of a woman’s arm by a trained health care provider. Sino-implant (II)/Levoplant is manufactured in China by Shanghai Dahua Pharmaceutical Co., Ltd. In June 2017, the product was prequalified by WHO. WHO prequalification has allowed additional donors to purchase the product for country programs; 1 million units were procured in 2018, for a total of 2.5 million units procured during the whole initiative.
Through this project, FHI 360 provided technical assistance to facilitate the global introduction of Sino-implant (II)/Levoplant, including conducting independent quality testing, negotiating public-sector price-ceiling agreements, supporting the WHO prequalification application process and working with distributors to secure national regulatory approvals. FHI 360 led clinical trials in China and the Dominican Republic, which supplemented existing clinical evidence, and collaborated on post-marketing surveillance studies in four countries with support from the Bill & Melinda Gates Foundation and the U.S. Agency for International Development (USAID). FHI 360 has worked in close coordination with a number of partners on the initiative, including DKT WomanCare, which became the global marketing partner for Dahua in 2018.