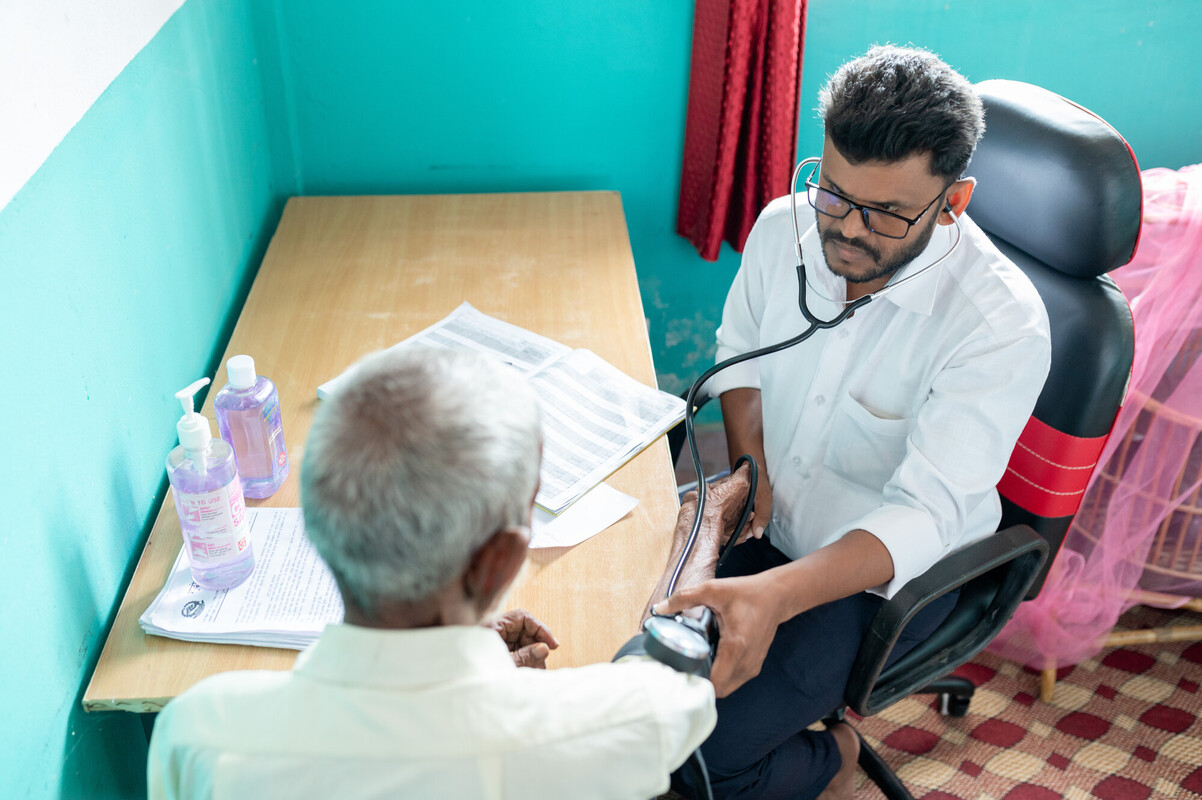
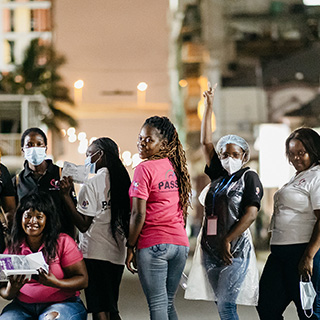FHI 360 serves as the Leadership and Operations Center for the HIV Prevention Trials Network (HPTN), a global, collaborative clinical trials network that brings together scientists, ethicists, community representatives and other partners to develop and test the safety and efficacy of interventions designed to prevent the acquisition and transmission of HIV.
HPTN’s groundbreaking studies have led to the global adoption of antiretroviral therapy for preventing HIV transmission (HPTN 052) and the approval of long-acting injectable cabotegravir for preventing HIV acquisition (HPTN 083 and HPTN 084).
FHI 360 facilitates and manages the network’s scientific agenda and research operations, from study development and implementation to publication of results. Our staff partner with investigators, clinicians and community representatives from clinical research sites in Africa, Asia, North America and South America on all aspects of the HPTN research program. We also work closely with the network’s sponsors, leaders, funders, pharmaceutical partners and institutional stakeholders, including the HPTN Statistical and Data Management Center, located at the Fred Hutchinson Cancer Center, and the HPTN Laboratory Center at Johns Hopkins University.
As the Leadership and Operations Center for HPTN, FHI 360 participates in and supports:
- Network leadership and governance.
- Research development, management and oversight.
- Clinical site preparation, training and technical assistance.
- Community engagement and research ethics programs.
- Communications, information management and publicity.
- Conference/event planning and management.
- Financial management and oversight.
For news, updates and more information about the HPTN, visit hptn.org and on social media (@HIVptn).