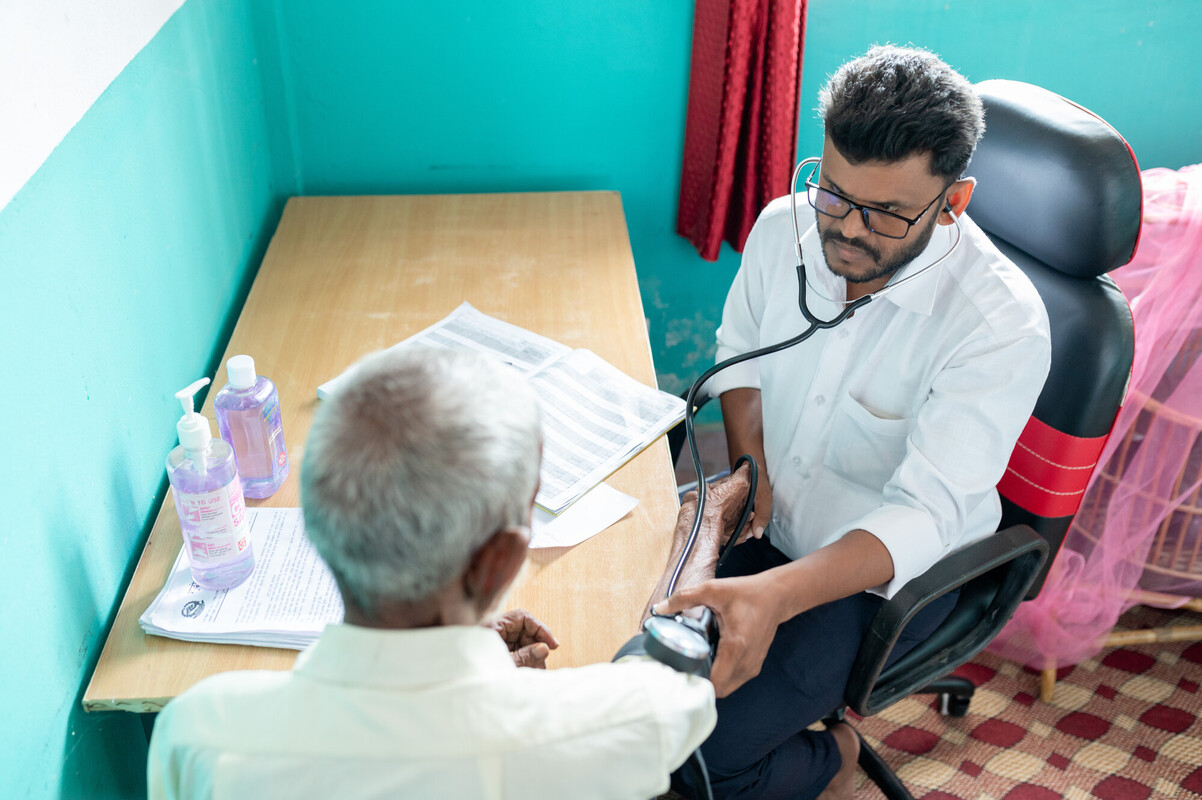
FHI 360 envisions a world where individuals have access to an expansive range of family planning (FP) methods to meet their changing needs throughout their reproductive lives. This vision drives the research agenda set forth under Envision FP, a contraceptive technology research project of the U.S. Agency for International Development.
With an overall goal to develop, introduce and expand understanding of contraceptive technologies to enhance choice and reduce unmet need, the Envision FP research agenda has three aims:
- Develop new methods to address significant gaps in the current method mix
- Refine existing products to enhance safety, use and acceptability
- Respond to product-related issues from the field that could affect procurement, provision or uptake
Research activities
The research portfolio targets multiple contraceptive methods, with activities spanning the spectrum of product development. Collaborating with strategic partners, FHI 360 scientists are:
- Developing a proof of concept and conducting acceptability research for a microneedle patch that delivers contraceptive hormones to women
- Evaluating whether a lower-dose version of subcutaneous depot-medroxyprogesterone acetate (SQ DMPA) is as effective as other contraceptive injectables currently on the market
- Conducting a mixed-methods study to determine barriers and facilitators to receiving high-quality, contraceptive implant removal services
- Providing technical assistance to the makers of a new, easy-to-use, postpartum inserter for intrauterine devices to support efforts to attain United Nations Population Fund/World Health Organization prequalification status and CE marking
- Supporting global introduction and evaluation of the levonorgestrel intrauterine system
- Hosting a database that lists potential interactions between contraceptive drugs and drugs from other therapeutic classes
- Identify ways that considerations related to noncontraceptive health benefits and contraceptive-induced menstrual changes can be integrated better into contraceptive research, development and introduction.
Responding to field-generated product concerns
When concerns about contraceptives arise in the field, loss of trust can lead to dramatic disruptions in procurement, provision and uptake. FHI 360’s rapid-response team identifies root causes, evaluates corrective action options, works with stakeholders to develop resolution plans and provides post-incident monitoring.
Work under Envision FP builds on FHI 360’s experience over four decades in the research, development and introduction of new contraceptive methods. Much of this work continues through the USAID-funded Innovate FP project. The project also leverages FHI 360’s Contraceptive Technology Innovation Initiative, a research grant funded by the Bill & Melinda Gates Foundation, and additional research activities funded by the foundation.
For more information about Envision FP, or to notify the rapid response team, please contact us.