Since the earliest days of the COVID-19 pandemic, FHI 360 has supported multifaceted prevention efforts, including developing vaccines and medicines, increasing vaccination coverage through social and behavioral interventions and direct service delivery, and building facility capacity for infection prevention and control.
Increasing global vaccination coverage
The global effort to vaccinate people against COVID-19 becomes increasingly critical as new variants of the virus emerge. From the earliest days of vaccine availability, FHI 360 has supported all areas of vaccine rollout, including training frontline workers, building logistical and operational capacities, tracking adverse reactions, monitoring delivery systems and working with government leaders to create and carry out national vaccination plans. FHI 360 also works to foster vaccine confidence and counter misinformation by conducting outreach campaigns that include traditional communication methods as well as peer-to-peer programs.
Key COVID-19 vaccination resources:
- Demand Creation and Advocacy for COVID-19 Vaccine Acceptance and Uptake Toolkit
- Ensuring COVID-19 Vaccination for People Living with HIV and Key Populations
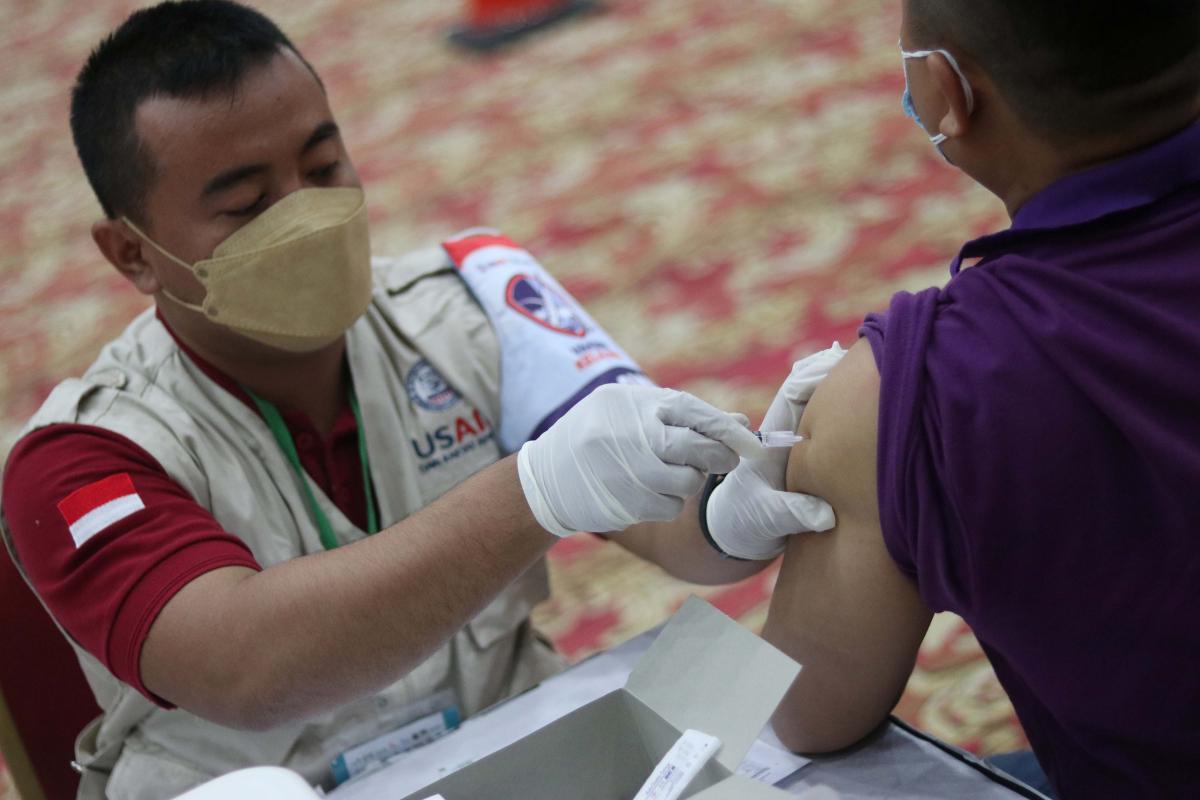
Increasing vaccine uptake through social and behavior change and risk communication and community engagement
FHI 360 offers global capabilities and technical expertise in social and behavior change (SBC) and risk communication and community engagement (RCCE) to support national efforts to introduce COVID-19 vaccines, create demand, counter misinformation, address vaccine hesitancy and create an enabling environment for sustained use of COVID-19 vaccines. FHI 360 partners with and strengthens the capacity of governments and local organizations to develop, implement, monitor and evaluate SBC and RCCE activities designed to encourage targeted populations to access vaccines, trust their effectiveness, believe they are relevant to them and have the self-efficacy and motivation to begin and complete a vaccine regimen when available. To support governments in the development and implementation of COVID-19 vaccine programs, FHI 360 developed the Demand Creation and Advocacy for COVID-19 Vaccine Acceptance and Uptake: Quick Start Guide. The guide and accompanying tools build on existing guidance from the World Health Organization and other global authorities to support users to design, implement, monitor and redesign plans for creating vaccine demand, strategies for vaccine advocacy, and RCCE interventions that address countries’ evolving needs.
Development of vaccines and medicines
As part of the U.S. government’s COVID-19 prevention agenda, FHI 360 partnered with the U.S. National Institutes of Health and leading pharmaceutical companies on two critical studies. The first was on a monoclonal antibody to prevent COVID-19 infection in senior nursing facilities, which suffered disproportionately high rates of infection compared to the general population. The second was on a combination of two monoclonal antibodies for prevention in households with a positive case. These two studies helped provide the scientific framework needed to support the emergency use authorization from the U.S. Food and Drug Administration for a COVID-19 vaccine.
Infection prevention and control
FHI 360 supports countries to assess gaps in health systems and train frontline health care workers to prepare for COVID-19 outbreaks, with a focus on robust infection prevention and control (IPC) initiatives. We provide technical assistance to develop and deploy IPC training packages across multiple tiers of the health care system.