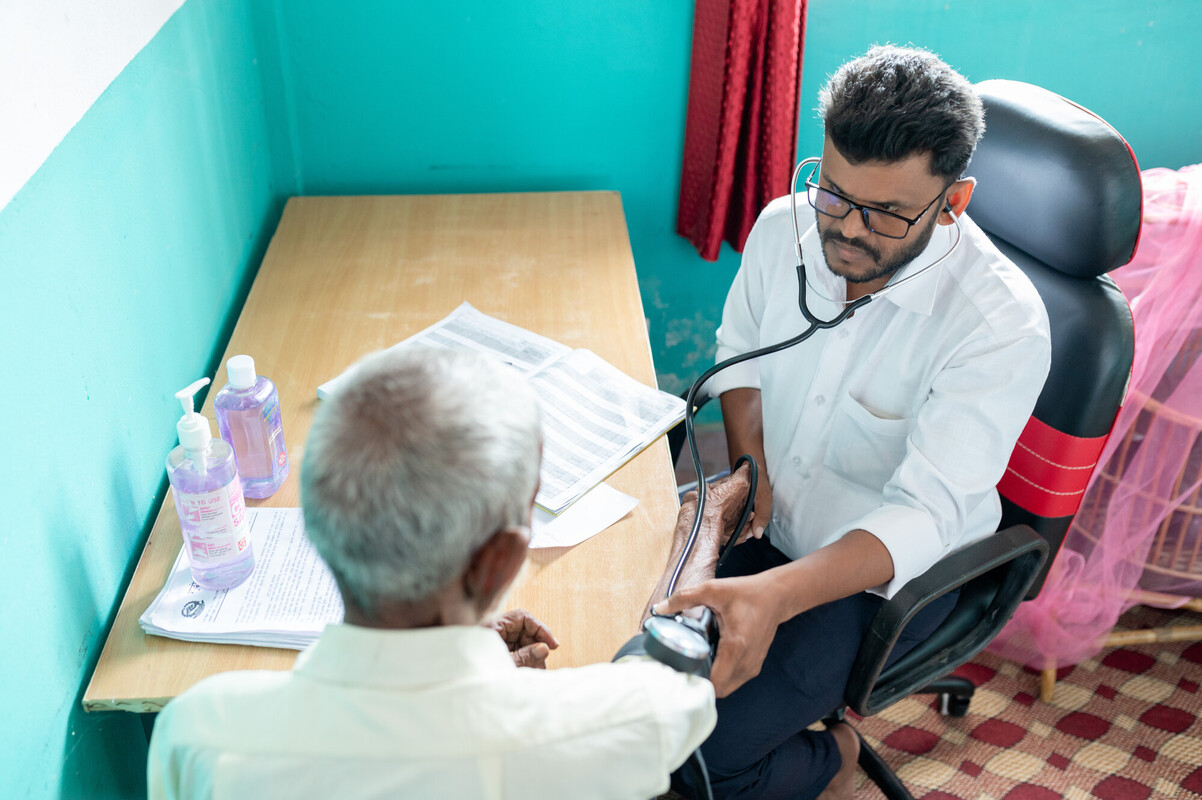
An estimated one-quarter of the world’s population has an inactive tuberculosis infection. In 2024, about 10.7 million people fell ill with an active TB infection, and 1.23 million people died from it. Although there is an effective treatment regimen for the world’s deadliest infectious disease, it requires patients to take multiple drugs daily for at least six months. Many experience prolonged and uncomfortable side effects.
Treatment adherence is a challenge everywhere. When people stop treatment early, they are more likely to relapse or develop multidrug-resistant TB. Treating this type of TB involves taking toxic drugs for an even longer period, with lower rates of success.
Patients, health workers and experts agree: a long-acting treatment option would be easier to follow, increasing completion rates and reducing the spread of TB. That’s why FHI 360 — with support from the Gates Foundation and in partnership with the Gates Medical Research Institute — is implementing a study to understand the preferences of potential end-users and providers of a hypothetical long-acting injectable TB treatment. We’re studying how this future option, which could treat millions, might be accepted by patients and adopted by health care workers, as well as the key factors that could support its uptake.
Here, FHI 360 Scientist Lara Lorenzetti, Ph.D., shares what the research has uncovered.
FHI 360: A long-acting TB medication doesn’t exist yet. Why should we do research on a drug before it’s been developed?
Lara: A drug can look “perfect” on paper and still be rejected by patients for a variety of reasons. We know from the uptick in injectable treatments in the HIV space and beyond that a long-acting injectable can solve some well-documented issues, notably by removing the burden of daily pill-taking. But a long-acting medication will only work if people are willing to use it.
The idea behind our research is to talk to potential end-users and health care providers and ensure their preferences are introduced early in the drug development process. This helps drug developers and policymakers avoid a situation where enormous amounts of time and resources are spent creating a treatment that health facilities aren’t equipped to administer and patients won’t use or be able to adhere to. And we can proactively design educational communications that address concerns about a treatment that is likely to be developed.
How is the research structured?
We are conducting a mixed-method study to understand provider and patient preferences for a hypothetical long-acting injectable TB treatment. We conducted a pilot study in the Philippines, and now we’re expanding and replicating the study in Indonesia and South Africa. We’re also analyzing data from a similar study in Tanzania.
The work is split into two pieces. The first includes qualitative key informant interviews with clinicians, government officials and members of medical organizations — professionals who can share provider- or systems-level perspectives related to a potential treatment. The second involves a survey with providers, patients in treatment and those who have completed treatment to understand what they might find acceptable or preferable about different options.
Drawing on literature reviews and expert feedback, we created three hypothetical scenarios involving an injectable TB treatment in some form. The first involves four injections during a one-time treatment. The second involves two injections per visit over a three-month period. And the third involves taking daily pills for one month, followed by two injections given two months apart.
We pitched these scenarios in the interviews and survey. The survey also included a discrete choice experiment to help us understand trade-offs participants might make. For example, is it more important to receive fewer shots per visit or to have treatment delivered nearby at a community health center? We knew from discrete choice experiments about HIV treatment that cost and efficacy are consistently ranked as the most important attributes. We told participants to assume these hypothetical treatment options were free and equally efficacious. This helped us learn about other key elements of a treatment regimen and its delivery that might influence whether a person starts or continues treatment.
Who participated in the study?
We interviewed 15 key informants in the Philippines and surveyed about 350 TB patients, survivors and providers. In Indonesia, South Africa and Tanzania, the goal is to reach roughly 20 key informants and 1,050 survey participants per country. The study seeks opinions from people with multidrug-resistant TB and those with drug-susceptible TB (which responds to first-line treatment). We selected facilities in urban and semi-urban areas for demographic range.
Ideally, when the treatment is developed, countries all over the world will have access to it. We tried to reach a diversity of groups to understand what will work for most, recognizing that people are complex and no one option will work for everyone.
What have you learned so far that was unexpected?
We initially expected that patients would prefer to go to a health facility once, even if it meant getting several shots in one visit. But that wasn’t what we found in the Philippines. People don’t want multiple deep muscle injections at once, especially if it might cause severe injection site pain. Instead, many are open to receiving fewer shots monthly for several months or even an initial phase of daily pills followed by periodic injections. And, of course, they want treatment available at their nearest health facility.
Providers and key informants saw the appeal of having patients come once, but they were also interested in having patients return multiple times so they could be monitored for adverse reactions.
In the coming months, we are eager to analyze data from Indonesia, South Africa and Tanzania, to see how patient and provider perspectives may support or differ from what we found in the Philippines.
What drew you to this work?
Millions of people, every day, deal with this disease. For many, it’s a death sentence. It’s motivating to participate in developing a product that could treat patients and truly fit within their lifestyles, preferences, budgets — to make a product that actually works for people. Because when lives are in the balance, we need to make sure that medication will work and can be used as intended. We can’t waste time or resources.
TB patients are not just “end-users.” They’re mothers, fathers, daughters, sons — people who have thoughts, feelings and preferences that need to be valued and considered when we make policy decisions. I’m driven by trying to elevate those voices and make sure that they are a part of the conversation.
This work is supported by the Gates Foundation and conducted in partnership with the Gates Medical Research Institute, Dartmouth Health, TB HIV Innovations and Clinical Research Foundation in the Philippines, the Aurum Institute in South Africa, the Center for Health Research at the University of Indonesia, and Muhimbili University of Health and Applied Sciences in Tanzania.
Evidence into Action is a recurring series exploring how organizations, communities and governments can make better decisions using data. To learn how FHI 360 can support your work through market research, please contact partneringwithus@fhi360.org.